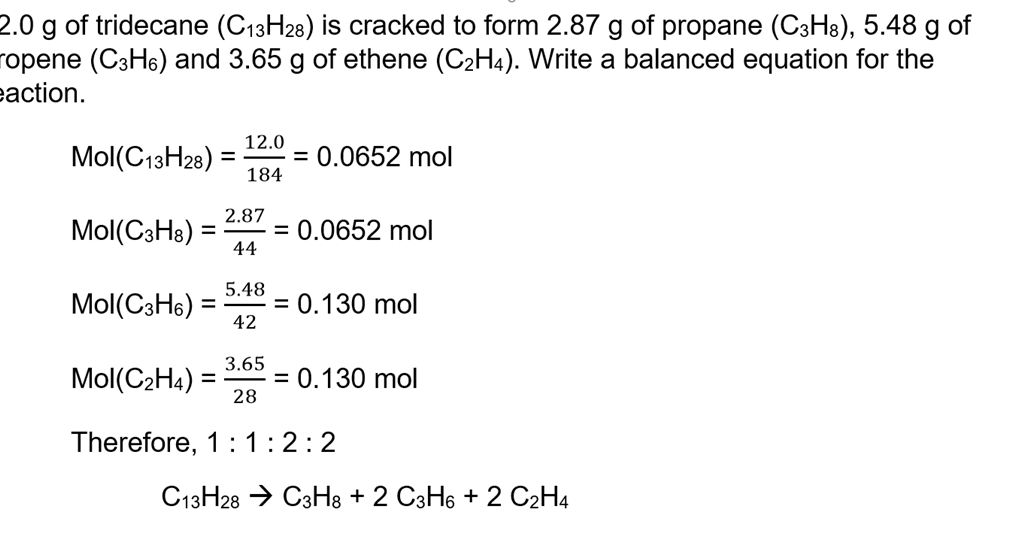
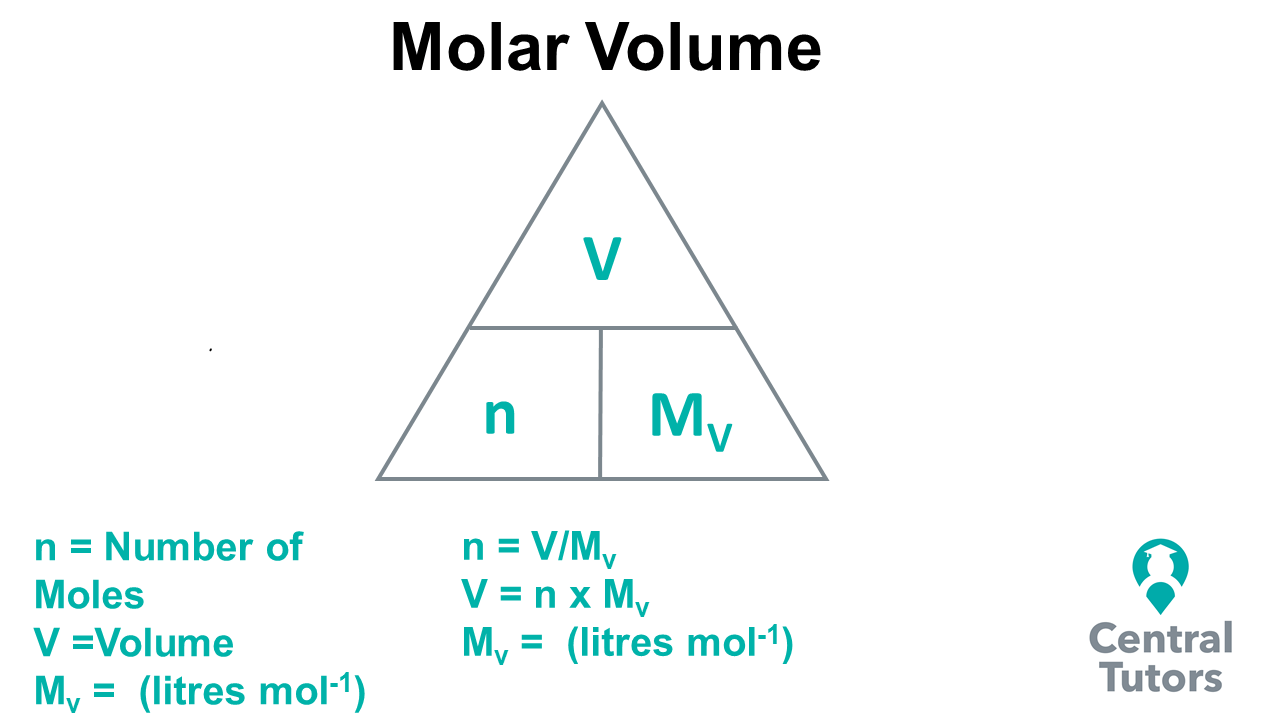
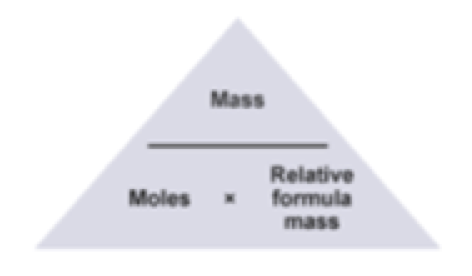
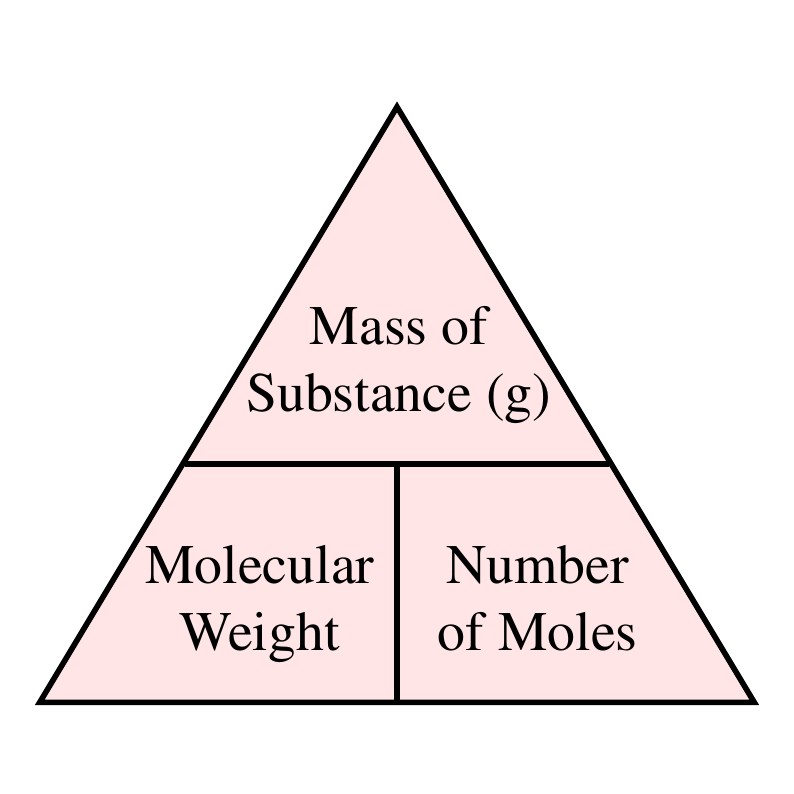
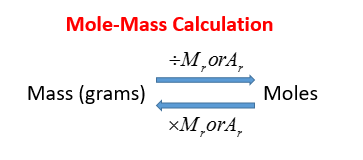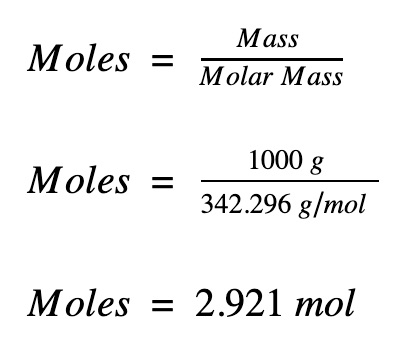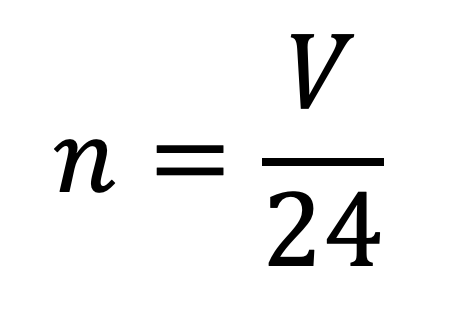The Mole Formula Triangle Or Pyramid With Avogadro Number Or Avogadro Constant NA = 6.022 × 1023 Mol − 1 Isolated On White. Relationship Between Moles, Number Of Particles, Avogadro Constant, Chemistry.

How To Solve Most Mole Calculation Questions – Part 1 | O Level Chemistry Tuition | Leading Chemistry Tuition Centre

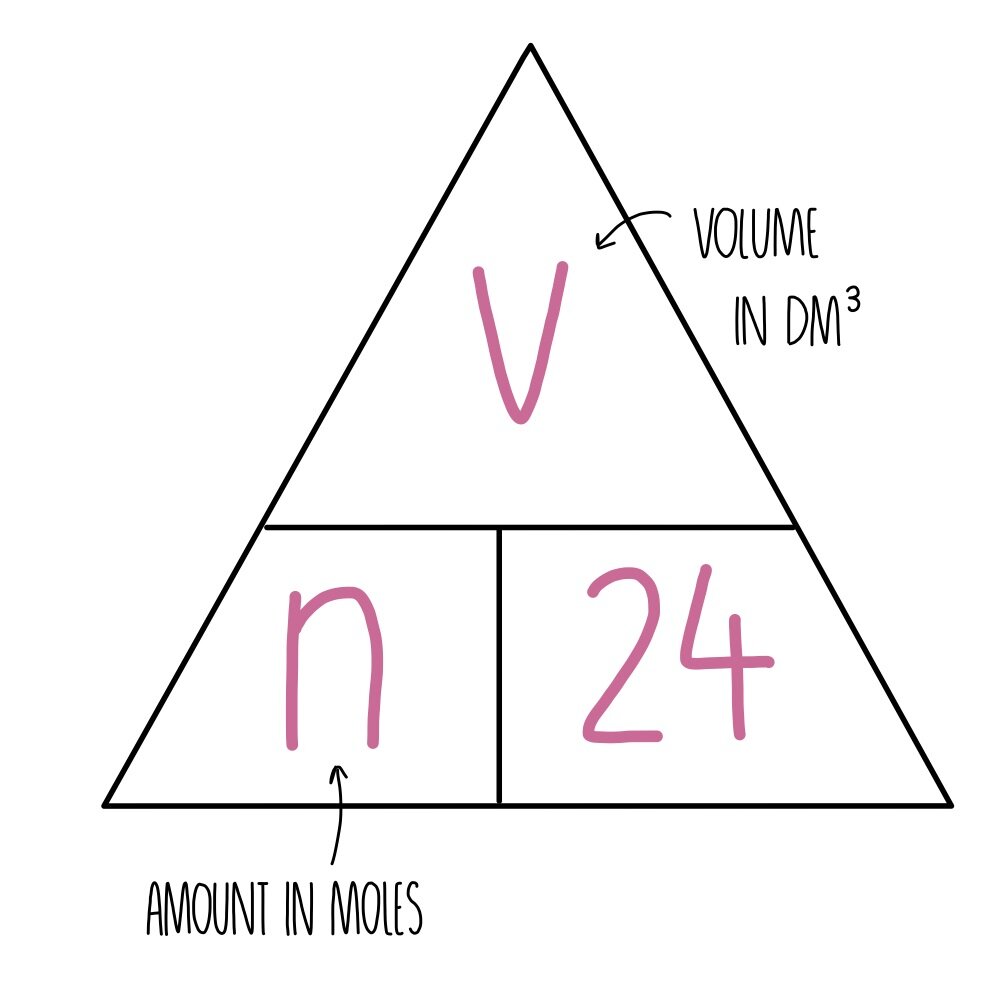
molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises
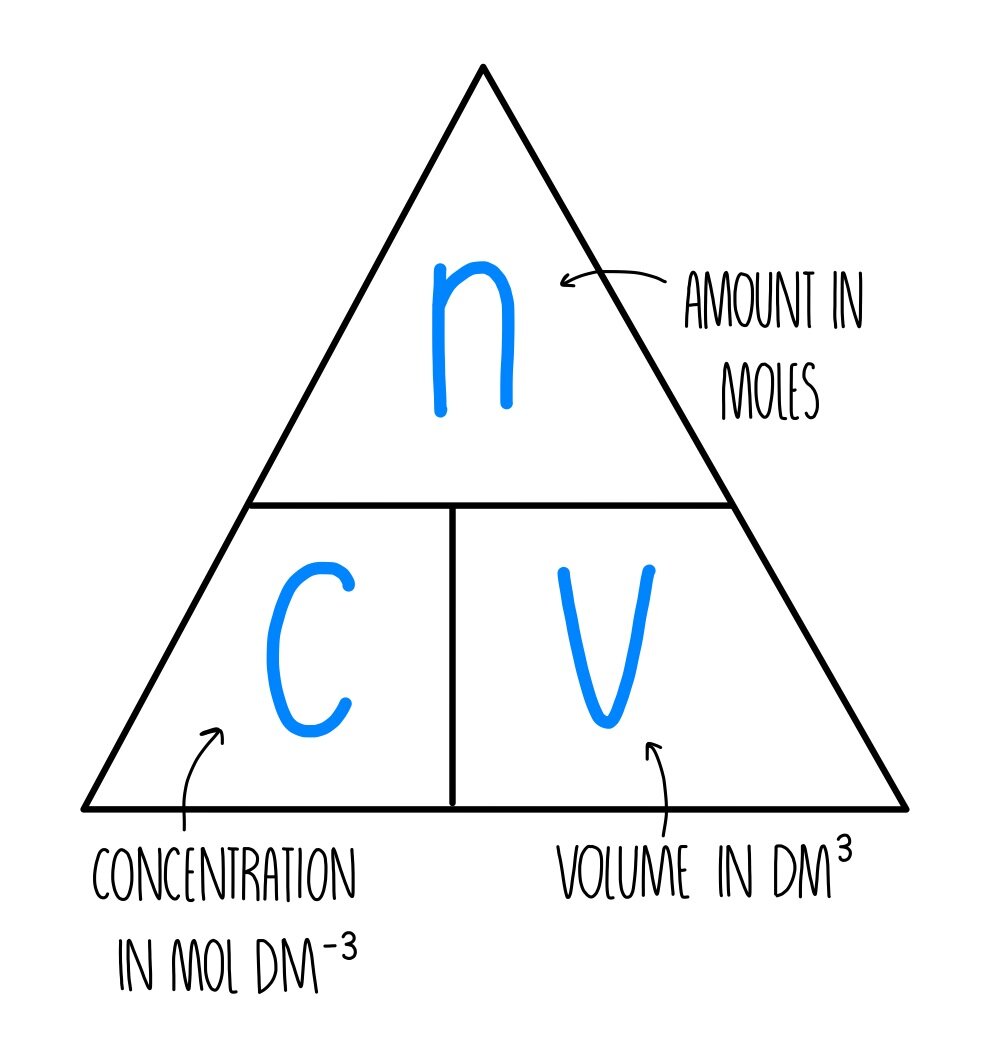
Mole calculations - The mole and concentration of solutions - National 5 Chemistry Revision - BBC Bitesize